When a hydrogen atom of an alkane is replaced by a halogen atom, the compound is haloalkane (monohaloalkane).
\underset{alkane}{R-H}\ \xrightarrow[+X]{-H}\ \underset{Haloalkane}{R-X}Classification

A. On the basis of attachment of halogen atom with different nature of carbon, haloalkane is classified into three categories:
1. Primary (1°) Haloalkane: Haloalkane in which halogen atom is attached with primary carbon is called primary haloalkane.

2. Secondary (2°) Haloalkane: Haloalkane in which halogen atom is attached with secondary carbon is called secondary haloalkane.

3. Tertiary (3°) Haloalkane: Haloalkane in which halogen atom is attached with tertiary carbon is called tertiary haloalkane.

B. On the basis of the number of halogen atoms, haloalkanes are classified into three categories:
1. Monohaloalkanes: They consists of only one halogen atom.
eg. CH3Cl, CH3CH2Cl
2. Dihaloalkane: They consists of two halogen atoms. They are further subdivided into two categories:
- Geminal dihalide: Those dihalogen compounds having the same types of halogen atom present on the same carbon atom. They are also called alkylidene dihalide. eg:

- Vicinal dihalide: Those dihalogen compounds having same types of halogen atom present on the adjacent carbon atom. They are also called alkylene dihalide. eg.

3. Polyhaloalkane: They consist of three or four halogen atoms.
eg. CHCl3, CCl4, Cl2CHCH2Cl, etc.
Nomenclature of haloalkane

Isomerism
Alkyl halide can show the following two isomerism:
- Chain Isomerism :

- Positional Isomerism:

General Methods of Preparation of haloalkanes:
1. From alkane: Alkane reacts with halogen in presence of sunlight or heat to give haloalkane.
\underset{}{R-H} + X_{2}\ \overset{h\nu}{\rightarrow}\ \underset{}{R-X} + HX\\ \underset{}{CH_{3}CH_{3}} + Cl_{2}\ \overset{h\nu}{\rightarrow}\ \underset{}{CH_{3}CH_{2}Cl} + HClNote: It proceeds through a free radical substitution mechanism.
2. From Alkene: Alkene reacts with hydrogen halide to give haloalkane.
\begin{align*} CH_{2}=CH_{2}+HX &\rightarrow CH_{3}CH_{2}X\\ CH_{2}=CH_{2}+HCl &\rightarrow CH_{3}CH_{2}Cl\\ \end{align*}Important Note:
Markovnikov’s rule
When an unsymmetrical reagent is added to an unsymmetrical alkene, the positive part of the reagent goes to the unsaturated carbon having a higher number of hydrogen. It takes place for HCl, HBr and HI. It proceeds through an electrophilic addition mechanism.
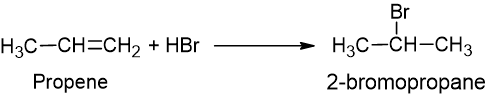
Anti-Markovnikov rule/peroxide effect/Kharash effect
When an unsymmetrical reagent(HBr) is added to an unsymmetrical alkene in presence of organic peroxide, bromine is added to the unsaturated carbon having more number of hydrogen. It proceeds through a free radical addition mechanism.
\underset{Propene}{CH_{3}CH=CH_{2}}\ + HBr\ \xrightarrow[peroxide]{benzoyl}\ \underset{1-bromopropane}{CH_{3}CH_{2}CH_{2}Br}3. From Alcohol:
i. Action with halogen acid: Alcohol reacts with halogen acid to give haloalkane.
\begin{align*} \underset{alcohol}{R-OH}+HX\ &\rightarrow\ R-X+H_{2}O\\ \underset{ethanol}{CH_{3}CH_{2}OH}+HCl\ &\xrightarrow[ZnCl_{2}]{anhy.} CH_{3}CH_{2}Cl+H_{2}O \end{align*}A mixture of HCl and anhydrous ZnCl2 is called Luca’s reagent. This reaction is called the grooves process.
\begin{align*} \underset{ethanol}{CH_{3}CH_{2}OH}+HBr\ &\xrightarrow[H_{2}SO_{4}]{conc.}\ \underset{bromoethane}{CH_{3}CH_{2}Br}+H_{2}O\\ \underset{ethanol}{CH_{3}CH_{2}OH}+HI\ &\rightarrow\ \underset{Iodoethane}{CH_{3}CH_{2}I}+H_{2}O \end{align*}- Reactivity of alcohol: 3°>2°>1°
- Reactivity of HX : HI > HBr > HCl
ii. Action with phosphorous halide:
\begin{align*} CH_{3}CH_{2}OH+PCl_{3}\ &\rightarrow\ CH_{3}CH_{2}Cl + H_{3}PO_{3}\\ CH_{3}CH_{2}OH+PCl_{5}\ &\rightarrow\ CH_{3}CH_{2}Cl + POCl_{3}+HCl \end{align*}iii. Action with thionyl chloride: Alcohol reacts with thionyl chloride in presence of pyridine to give chloroalkane. This reaction is called Dargen’s reaction. Here, HCl is absorbed by pyridine and SO2 escapes out. The only chloroalkane is obtained. So, SOCl2 is the best chlorinating agent.
\begin{align*} R-OH+\underset{thionyl\ chloride}{SOCl_{2}} &\xrightarrow[]{pyridine} R-Cl+SO_{2}+HCl\\ CH_{3}CH_{2}OH+SOCl_{2} &\xrightarrow[]{pyridine} CH_{3}CH_{2}Cl+SO_{2}+HCl \end{align*}Physical Properties:
- Physical state, color, odour: CH3Cl, CH3Br, and C2H5Cl are colorless gases having characteristics odour, members up to C18 are sweet smelling colorless liquid and higher members are odourless solid.
- Solubility: They are insoluble in water but soluble in organic solvents like benzene, ether, chloroform, etc.
- Boiling point:
- For the same halogen atom, the boiling point increases with an increase in a carbon atom.
CH3Cl < CH3CH2Cl < CH3CH2CH2Cl - For the same alkyl group, the boiling point increases with an increase in size.
CH3Cl < CH3Br < CH3I - For isomeric haloalkane, the boiling point increases with a decrease in branching or an increase in surface area.

Chemical Properties:
Alkyl halides are highly reactive aliphatic compounds due to the polar nature of carbon to halogen bond. The reactivity of alkyl halide is in the order: R-Cl < R-Br < R-I
A. Nucleophilic Substitution Reaction
In alkyl halides, the carbon to halogen bond is polar since halogen is more electronegative than carbon. Therefore nucleophile attacks the carbon bearing the halogen atom to give a substitution reaction. A substitution reaction initiated by a nucleophile is called a nucleophilic substitution reaction.
\underset{substrate}{R-X}+\underset{reagent}{Nu^{-}}\xrightarrow[]{solvent}\underset{product}{R-Nu}+\underset{leaving\ group}{X^{-}}Alkyl halides can undergo nucleophilic substitution reaction by following two mechanisms.
1. SN2 reaction
It stands for bimolecular nucleophilic substitution reaction. When the rate of nucleophilic substitution reaction depends on both the concentration of alkyl halide(substrate) and nucleophile, it is called SN2-reaction. It follows second order kinetics. It completes in one step through the formation of transition state having partial cleavage bond with halogen and partial bond formation with a strong nucleophile.

As the nucleophile starts to form a bond with the substrate, leaving group also starts to break the bond with the substrate and finally leaving group leaves to give the product. This means the reaction takes place in a single step without any intermediate. Due to the backside attack of the nucleophile, the product is obtained with complete inversion of configuration like an inversion of an umbrella. This is called Walden inversion.
Rate of reaction : k [substrate][nucleophile]
Order of reactivity of haloalkane for SN2-reaction is 1°>2°>3°.
2. SN1 reaction
It stands for unimolecular nucleophilic substitution reaction. When the rate of nucleophilic substitution reaction depends on the only concentration of alkyl halide(substrate), it is called SN1-reaction. It follows first-order kinetics. It completes in two steps:
Step I-Formation of carbocation:
Alkyl halide ionizes to give carbocation. This step is slow and hence it is called the rate-determining step(rds).

Step II- Attack of nucleophile:
Nucleophile attacks the carbocation to give a product. Since carbocation is always planar, nucleophile attacks from either side to give products. Hence, the product obtained is inversion as well as retention of configuration.

Rate of reaction = k[substrate]
Order of reactivity of SN1 reaction is 3°>2°>1°
Difference between SN1 and SN2 reaction
| SN1 reaction | SN2 reaction |
| Unimolecular reaction | Bimolecular reaction |
| Two steps mechanism | Only one step mechanism |
| The rate depends on the concentration of alkyl halide | The rate depends on the concentration of alkyl halide and alkali. |
| Carbocation intermediate is formed | Takes place through a transition state |
| Mostly occurs in 3° alkyl halide | Mostly occurs in 1° alkyl halide |
| A racemic mixture is formed | Walden inversion takes place |
| Favourable in a protic solvent | Favourable in an aprotic solvent |
| Order of reactivity: 3°>2°>1° | Order of reactivity: 1°>2°>3° |
| Rearrangement may take place | No rearrangement takes place |
i. Action with KOH and NaOH: Haloalkane reacts with aqueous KOH and NaOH to give alcohol.
\begin{align*} R-X + KOH(aq)&\rightarrow R-OH + KX\\ CH_{3}CH_{2}Cl + KOH(aq)&\rightarrow CH_{3}CH_{2}OH + KCl \end{align*}ii. Action with potassium sulphide(K2S):
\begin{align*} 2R-X + K_{2}S&\rightarrow \underset{alkyl\ thioalkane}{R-S-R} + 2KX\\ CH_{3}CH_{2}Cl + K_{2}S&\rightarrow \underset{ethyl\ thioethane}{CH_{3}CH_{2}-S-CH_{2}CH_{3}} + KCl \end{align*}iii. Action with alcoholic sodium alkoxide: Haloalkane is reacted with sodium or potassium alkoxide to give ether. This reaction is called Williamson’s synthesis.

iv. Action with alcoholic KCN or NaCN: Haloalkane is treated with alcoholic KCN or NaCN to give alkanenitrile (alkyl cyanide).
\begin{align*} R-X+KCN(alc.)&\rightarrow \underset{Alkanenitrile}{R-CN}+KX\\ CH_{3}CH_{2}Cl+KCN(alc.)&\rightarrow \underset{Propanenitrile}{CH_{3}CH_{2}CN}+KCl \end{align*}
v. Action with alcoholic AgCN: Haloalkane is reacted with alcoholic AgCN to give isonitrile (isocyanide).

Q: When ethyl iodide reacts with KCN, ethyl cyanide is formed but with AgCN, ethyl isocyanide is formed. Explain.
A: In the case of KCN, there is an ionic bond between K+ and CN-. Cyanide ion has an independent existence in which there are two nucleophilic centres (C and N) but nucleophilic power of C is more than N. So, -CN group is introduced. Hence, cyanide is formed.
But in the case of AgCN, there is a covalent bond between Ag and CN that doesn’t ionize due to which nitrogen acts as a nucleophilic centre. Hence, isocyanide is formed.
vi. Action with alcoholic potassium nitrite: Haloalkane is treated with alcoholic KNO2 to give alkyl nitrite.
R-X + KNO_{2}(alc)\rightarrow \underset{Alkyl\ nitrite}{R-ONO} + KXvii. Action with alcoholic AgNO2: Haloalkane is treated with alcoholic AgNO2 to give nitroalkane.
\begin{align*} R-X + AgNO_{2}(alc)&\rightarrow \underset{Nitroalkane}{R-NO_{2}} + AgX\\ CH_{3}CH_{2}Cl + AgNO_{2}(alc)&\rightarrow \underset{Nitroethane}{CH_{3}CH_{2}NO_{2}} + AgCl \end{align*}*Ambident nucleophile: The nucleophile in which more than one atom acts as a nucleophilic site to attack the electron-deficient centre is called ambident nucleophile. eg. NO2–, CN–.
viii. Action with ammonia: Alkyl halide reacts with alcoholic ammonia solution to give primary amine.
R-X+\underset{excess}{NH_{3}}\rightarrow \underset{1^{\degree} amine}{R-NH_{2}}+HXIf alkyl halide is excess, primary, secondary, and tertiary amine with quaternary ammonium salt is formed. This is called the Hoffman ammonolysis process.

2. Elimination reaction (Dehydrohalogenation)
Haloalkane is boiled with an alcoholic KOH solution to give an alkene.

H-atom is lost from β-carbon. So, it is also called β-elimination. If there is a possibility of forming two different alkenes, the more substituted alkene is formed as a major product. This is called the saytzeff rule.

3. Action with metals
i. Action with sodium: Haloalkane is reacted with sodium metal in presence of dry ether to give alkane having a double number of carbon atoms is formed. This reaction is called the Wurtz reaction.

Note:
- It is not suitable for methane formation.
- It is not suitable for the formation of alkane having odd number of carbon. It is only suitable for alkane having even number of carbon because during formation of alkane having odd number of carbon, mixture of alkanes are obtained.
4. Reduction
\underset{Haloalkane}{R-X}\xrightarrow[LiAlH_{4}]{H_{2}/Ni\ or\ Pt\ or\ Pd} \underset{Alkane}{R-H}*Reducing agents: H2/Ni, H2Pd, H2/Pt, Sn/HCl, Fe/HCl, LiAlH4, NaBH4, Na/C2H5OH, Ni-Al/NaOH


