Phenols are compounds that have a hydroxyl group (-OH) bonded directly to an aromatic ring. They are represented by the general formula Ar-OH where Ar is phenyl, substituted phenyl or some other aryl group. When the hydroxy group is bonded to a carbon of the side chain, Ar-CH2-OH, the compound is termed as aromatic alcohol.

Classification and nomenclature
According to the number of -OH group connected in the aromatic ring, phenols are classified into:
1. Monohydric phenol

2. Dihydric phenol

3. Trihydric phenol

Preparation of phenol:
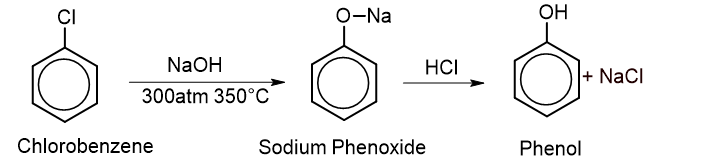
1. From chlorobenzene: Chlorobenzene is heated with NaOH at 350 °C and 300atm to give sodium phenoxide which on acidification gives phenol. This reaction is called the Dow process.
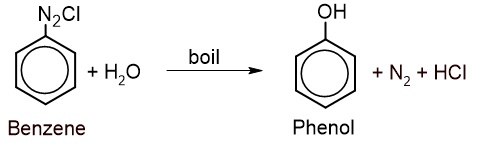
2. From benzene diazonium chloride: Benzene diazonium chloride is boiled with water to give phenol.
3. From benzene sulphonic acid: Sodium benzene sulphonate is heated with NaOH at high temperature followed by acidification gives phenol.

Physical properties:
- It is a white crystalline solid having sweet odour.
- It is partially soluble in water.
- Its melting point is 40.5 °C.
- Phenol with 5% water is commonly called carbolic acid.
Chemical Properties:
1. Acidic nature of phenol: Acid strength of an acid depends upon the stability of the conjugate base. With the increase in the stability of the conjugate base, acidic strength increases. Phenoxide ion (the conjugate base of phenol) is stabilized by resonance. So its acid (phenol) is acidic in nature.

\underset{Alcohol}{R-O-H}\rightleftharpoons \underset{Alkoxide\ ion}{R-O}^{-}+H^{+}Alkoxide ion (the conjugate base of alcohol) is not stabilized by resonance. Hence phenol is more acidic than alcohol.
Due to charge separation in phenol, phenoxide ion is more stable than phenol.
Phenol > Methanol > Water > Ethanol
Decreasing order of acidic strength
2. Action with Na and NaOH:

Effect of substituents on the acidic strength of phenols
The acidic strength of phenol is affected by the presence of substituents in the ring.
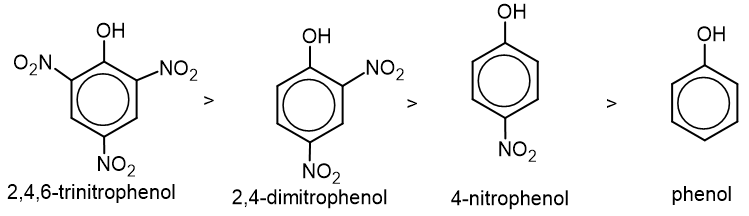
- With the introduction of electron withdrawing groups like -NO2, -X, -CHO, etc., acidic strength increases.

- With the introduction of electron releasing groups like -NH2, -R, -OR, etc, acidic strength decreases.
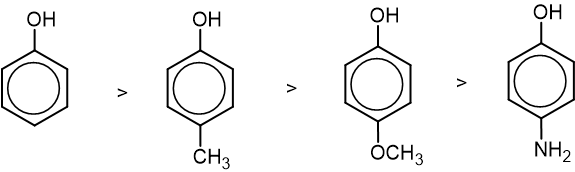
- The effect of any substituent is more significant when it is present in ortho or para position than in meta position.

- The increase or decrease in the number of substituents also affect the acidity.
3. Action with zinc: Phenol is heated with zinc dust to give benzene.

4. Action with ammonia: Phenol reacts with ammonia in presence of anhydrous ZnCl2 to give aniline.

5. Action with benzene diazonium chloride: Phenol reacts with benzene diazonium chloride in an alkaline medium to give p-hydroxy azobenzene which is also known as an azo dye. This reaction is called a coupling reaction.

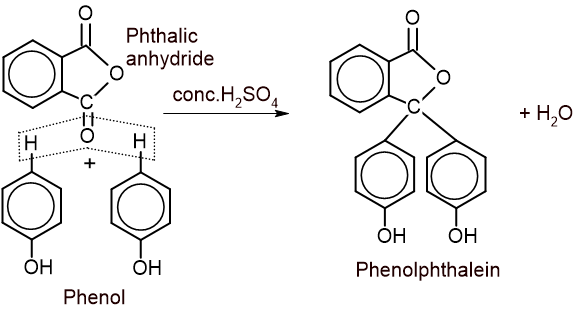
6. Action with phthalic anhydride: Phenol is heated with phthalic anhydride in presence of conc. H2SO4 to give phenolphthalein.
7. Acylation: Phenol reacts with acid chloride or acid anhydride to give ester.

8. Kolbe’s reaction: Sodium phenoxide is heated with CO2 at 125 °C and 4-7 atm followed by acidification to give salicylic acid. This reaction is called Kolbe’s reaction.

9. Reimer-Tiemann’s reaction: Phenol is heated with chloroform in presence of aq.KOH followed by acidification gives salicyaldehyde as a major product. This reaction is called the Reimer Tiemann reaction.

Electrophilic substitution reaction
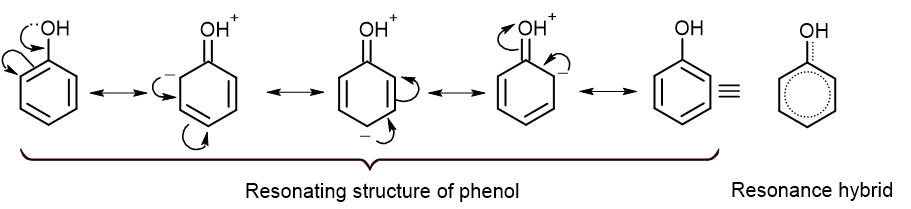
From the above resonating structure, it is clear that the -OH group increases the electron density at ortho and para positions. So, incoming electrophile prefers to attack at ortho and para position. Hence, the -OH group is an ortho para directing group.
1. Bromination (test of phenol): Phenol reacts with aqueous bromine to form a white precipitate of 2,4,6-tribromo phenol.

Due to the strong activating nature of the -OH group, the reactivity of phenol is very high.
Monobromophenol is obtained by reacting phenol with bromine in presence of less polar solvents like CS2 or CCl4 at low temperatures.

2. Nitration: Phenol reacts with conc. HNO3 to form 2,4,6-trinitrophenol.

Phenol reacts with dil. HNO3 to give mononitrophenol.

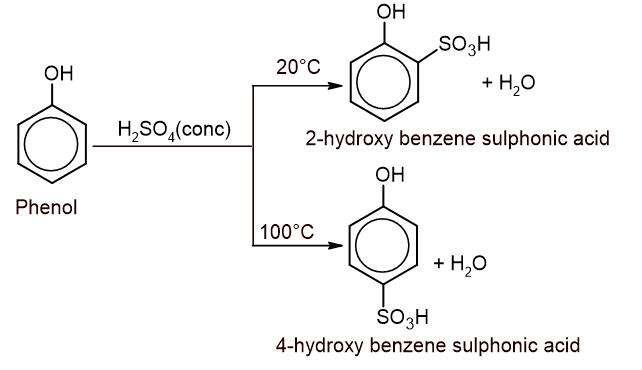
3. Sulphonation:
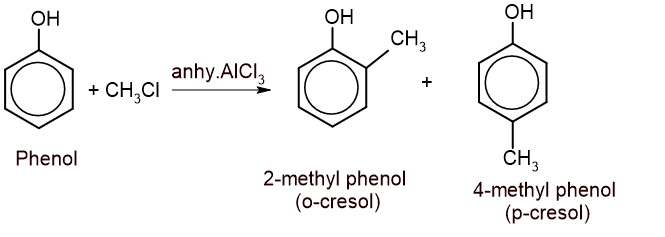
4. Friedel-Craft alkylation:
Laboratory test of phenol
1. Ferric chloride test: Phenol reacts with FeCl3 solution to give green, blue, violet, or red coloured water-soluble complex.

2. Bromine water test: Phenol reacts with bromine water to form white ppt. of 2,4,6-tribromo phenol.

3. Libermann test: Phenol gives red colour on reacting with nitrous acid. This red colour is changed into blue colour by adding NaOH.

Uses
- In the manufacture of dyes, drugs, explosives, etc.
- In the manufacture of bakelite.
- In the preparation of picric acid and phenolphthalein.
- As disinfectant and germicides.
Some Important Questions
- How do you obtain phenol from i.chlorobenzene ii. benzene diazonium chloride
- Why is phenol more acidic than ethanol?
- What happens when
i. Phenol is allowed to react with phthalic anhydride?
ii. Phenol is nitrated with dil. HNO3?
iii. Phenol is treated with bromine water?
iv. Chlorobenzene is heated with NaOH at high temperature and pressure.
v. Phenol is heated with chloroform in presence of aqueous alkali and the product is acidified? - Draw the resonating structure of i. Phenol ii. Phenoxide ion.
- Why -OH group in phenol is not meta-directing group?
- Which of the following has higher boiling point: o-nitrophenol or p-nitrophenol
- Why phenol is insoluble in aqueous sodium carbonate but picric acid is soluble?
- Phenol is nitrated easily but benzene cannot, Why?
- Write the reaction between i. phenol and aq. Br2 ii. Phenol and conc. HNO3
- How would you convert phenol into i. Aspirin ii. Salicylic acid iii. Toluene iv. Chlorobenzene v. Aniline vi. Ester
- Identify A and B in the following chemical equation.
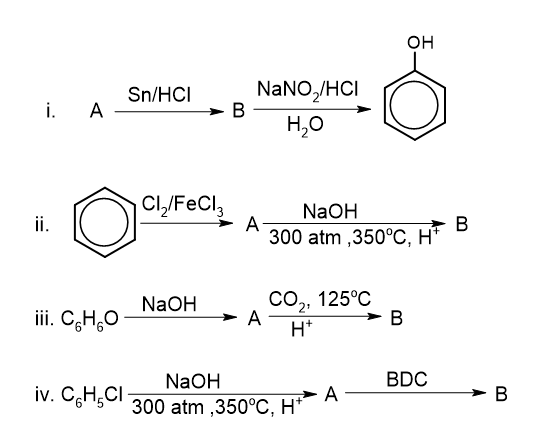
- How would you distinguish ethanol and phenol? Write with two reactions.
- How would you know if a given hydroxyl compound is an alcohol or phenol?
- Show your familiarity with Kolbe’s reaction.
- Arrange the following in decreasing order of their acidic strength
i. p-methoxy phenol ii. p-nitro phenol iii. phenol

