Nomenclature of organic compounds
1. Common system: In the early stage of the development of organic compounds, the organic compounds are named on the basis of their source of origin called a common system. Common names have many defects that are individual and unsystematic with the synthesis of a large number of organic compounds.
2. IUPAC system: Towards the end of the 19th century, for the systematic naming of organic compounds, the International Union of Pure and Applied Chemistry (IUPAC) is taken as standard and accepted all over the world known as the IUPAC system.
According to the IUPAC system, the name of organic compounds generally consists of the following parts.
Prefix + word root + primary suffix + secondary suffix
1. Prefix
It indicates the substituents or branching in the longest carbon chain.
| Substituent | Prefix | Substituent | Prefix |
| -CH3 | Methyl | -F | Fluoro |
| -CH2CH3 | Ethyl | -Cl | Chloro |
| -OCH3 | Methoxy | -Br | Bromo |
| -OCH2CH3 | Ethoxy | -I | Iodo |
| -NO2 | Nitro | -CN | Cyano |
| -NH2 | Amino | -OH | Hydroxy |
2. Word Root
It indicates the number of the carbon atom that is present in the longest carbon chain.
| Number of Carbon | Word Root | Number of Carbon | Word Root |
| 1 | Meth- | 5 | Pent- |
| 2 | Eth- | 6 | Hex- |
| 3 | Prop- | 7 | Hept- |
| 4 | But- | 8 | Oct- |
3. Primary Suffix
It indicates the nature of the Carbon to Carbon bond in the longest carbon chain.
| Nature of C to C bond | Primary Suffix |
| C–C (single bond) | -ane |
| C=C (double bond) | -ene |
| C≡C (triple bond) | -yne |
| C=C and C≡C (both single & double bond) | -en-yne |
4. Secondary Suffix
It indicates the main functional group that is present in the compound.
| Class | Sec. suffix | Class | Sec. suffix |
| Alcohol | -ol | Acid amide | -amide |
| Aldehyde | -al | Ester | -oate |
| Ketone | -one | Amine | -amine |
| Carboxylic acid | -oic acid | Cyanide | -nitrile |
| Acid chloride | -oyl chloride | Thioalcohol | -thiol |
| Acid anhydride | -oic anhydride |
Rules for IUPAC nomenclature of alkanes
1. Selection of longest carbon chain (parent chain): The chain having the maximum number of carbon is considered as the parent chain.

2. Numbering of carbon chain: If a substituent is present in the parent chain, the number of carbon is numbered from that end which gives the lowest number to the substituent.

i) If more than one substituent is present in the parent chain, numbering is done that gives the lowest possible sum.

ii.) Substituents are written in alphabetical order: Here e comes first and then the m in alphabetical order. So, ethyl comes first and then methyl while naming.
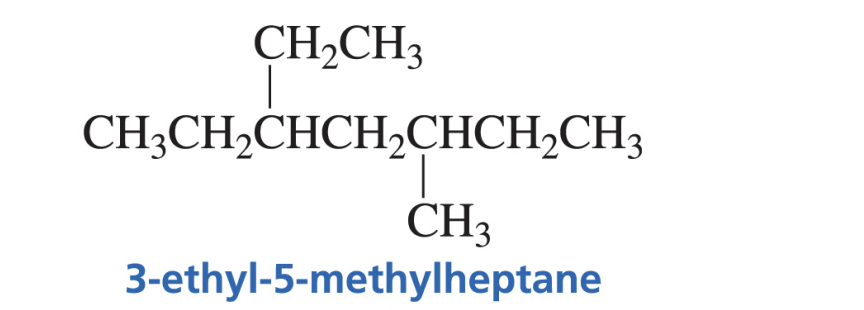
Examples of Nomenclature of Alkane

Examples of Nomenclature of Alkene

Examples of Nomenclature of Alkyne

Note: If double and triple bonds are present together, the naming of the compound will be as follows:

Haloalkane Naming Examples

Alcohol Naming Examples

Ether Naming Examples

Nitroalkane Naming Examples

Amine Naming Examples

Cyanide and Isocyanide Naming Examples

Aldehyde and Ketone Naming Examples

Carboxylic acid and acid chloride Naming Examples

Amide and Ester Naming Examples

Acid Anhydride Naming Examples

Some Additional Examples

Polyfunctional rule
If the parent chain contains more than one different functional group, the number of carbon atoms is counted from the end that gets the lowest number to the functional group of higher priority.
Order of priority is given as:
-COOH>-SO3H>-COOR>-COCl>CONH2>-CHO>-CN>-CO>-OH-NH2>C=C>-C C->-O->-NO2>-X>-R
Some Examples of Polyfunctional Compound


Some Important Questions
1. Write the IUPAC name of the following compounds.


2. Write the structure of the following compounds.



