NEB Grade 12 Chemistry 2082 Question Paper Solutions
This post contains the Grade 12 Chemistry 2082 Question Papers solutions. Includes Multiple Choice questions with answers, Short Questions with answers, Long Questions with answers, numerical problems and solutions. NEB Chemistry Past Question papers.
Group ‘A’ [11×1=11]
Rewrite the correct option of each question in your answer sheet.
1. To estimate the chloride ion present in tap water, volumetric analysis has been commonly used in the laboratory. Which of the following solutions can be used for the above mentioned titration?
A) BaCl2
B) NaOH
C) K2Cr2O7
D) AgNO3
2. The half-life of a certain reaction is 50 seconds, and it remains constant even if the initial concentration of the reaction is doubled. What is the order of the reaction?
A) Zero-order reaction
B) First-order reaction
C) Second-order reaction
D) Pseudo-first order reaction
3. Gibbs-Helmholtz equation is represented as ∆G = ∆H-T∆S. For what value of ∆G will the reaction be spontaneous?
A) Positive
B) Zero
C)Negative
D) Infinite
4. Standard reduction potential values for P, Q, R and S are +0.34V, -0.76V, +0.0V and -3.05V, respectively. What is the correct order of their reducing power?
A) P>R>Q>S
B) S>R>Q> P
C) Q>P>S>R
D) S>Q>R>P
5. A deep blue complex is formed when conc. ammonium hydroxide is added to an aqueous solution of CuSO4. Which of the following formulae represents the complex?
A) [Cu(NH3)4]2+
B) [Cu(NH3)4]+
C) [Cu(NH3)2]2+
D) [Cu(NH3)6]2+
6. Which of the following is the maximum possible oxidation state of the transition metals in the 3d series?
A) +3
B) +6
C) +7
D) +8
7. Which of the following are organometallic compounds?
i) CH3MgI
ii) Ni(CO)4
iii) C4H9Li
iv) C2H5ONa
v) (C4H9)2SnCl2
A) i)and ii) only
B) i), ii) and iii) only
C) i), ii), iii) and iv) only
D) i), ii), iii) and v) only
8. The organic compound (X) boiled with water gives phenol. What is the compound (X)?
A) Chlorobenzene
B) Benzenediazonium chloride
C) Benzenesulphonic acid
D) Aniline
9. Which of the following compounds gives a positive carbylamine reaction?
A) (CH3)2NH
B) (CH3)3N
C) (CH3)4N+
D) CH3NH2
10. A compound ‘Z’ with molecular formula C3H4O4 is heated to produce compound ‘Y’ and CO2 gas. Heating of ‘Y’ with P2O5 forms compound ‘X’ and a water molecule. Identify the compound ‘X’
A) Ethanoic acid
B) Ethanoic anhydride
C) Methanoic anhydride
D) Ethanal
11. A sequence of reactions is represented as:

Predict the name of the reaction that converts (B) to (C).
A) Benzoin condensation
B) Perkins’ condensation
C) Aldol condensation
D) Claisen condensation
Group ‘B’
12. Define instantaneous rate of reaction. Write down the rate expressions of the following reaction.
2N2O5(g) → 4NO2(g) + O2(g)
If 2.24 litres of O2 at NTP is produced in 30 minutes, what is the rate of disappearance of N2O5(g)?
Answer: Instantaneous rate of reaction is defined as the change in concentration of reactant or product at a particular instant or time. It is denoted by dx/dt.
For a general reaction:
aA + bB → cC + dD
\displaystyle
\frac{dx}{dt}
=
-\frac{1}{a}\frac{d[A]}{dt}
=
-\frac{1}{b}\frac{d[B]}{dt}
=
+\frac{1}{c}\frac{d[C]}{dt}
=
+\frac{1}{d}\frac{d[D]}{dt}Now, Rate expression:
\displaystyle
\frac{dx}{dt}
=
-\frac{1}{a}\frac{d[A]}{dt}
=
-\frac{1}{b}\frac{d[B]}{dt}
=
+\frac{1}{c}\frac{d[C]}{dt}
=
+\frac{1}{d}\frac{d[D]}{dt}\text{Rate expression:}\\
\text{Rate} = -\frac{1}{2}\frac{d[N_2O_5]}{dt}
= +\frac{1}{4}\frac{d[NO_2]}{dt}= \frac{d[O_2]}{dt}
\displaystyle
\begin{aligned}
\frac{d[O_2]}{dt}
&= \frac{2.24}{22.4 \times 30 \times 60}\ \text{mol s}^{-1} \\
&= 5.5 \times 10^{-5}\ \text{mol s}^{-1}
\end{aligned}\displaystyle
\begin{aligned}
\text{We have:}\\ \quad
\frac{1}{2}\frac{d[N_2O_5]}{dt}
&= \frac{d[O_2]}{dt} \\
\text{or,}\quad
\frac{d[N_2O_5]}{dt}
&= (5.5 \times 10^{-5})\times 2 \\
&= 1.1 \times 10^{-4}\ \text{mol s}^{-1}
\end{aligned}
The rate of disappearance of N2O5(g) is 1.1 x 10-4 mol sec-1.
13. Define standard heat of formation. Calculate the heat of formation of acetic acid from the following data:
C(s) + O2(g) → CO2(g), ∆ H = -99 Kcal/mol
H2(g) + 1/2O2(g) → H2O(l), ∆ H = -68 Kcal/mol
CH3COOH(l) + 2O2(g) → 2CO2(g) + 2H2O(l), ∆ H = -208 Kcal/mol
Answer: The enthalpy change (ΔH⁰f) when one mole of a compound is formed from its elements in their standard states under standard conditions (298 K, 1 atm, and 1 M concentration for solutions) is called the standard heat of formation.
2C(s) + 2H2(g) + O2(g) → CH3COOH(l), ∆ H = ?
We know that,
∆H = ∆H(product) – ∆H(reactant)
or, -208 = [2 x ∆H(CO2) + 2 x ∆H(H2O)] – [∆H(CH3COOH) + ∆H(O2)]
or, -208 = [2 x (-99) + 2 x (-68)] – [∆H(CH3COOH) + 0]
or, -208 = [-198 – 136] – ∆H(CH3COOH)
∆H(CH3COOH) = -126 Kcal/mol
‘OR’
You are given the standard reduction potentials:
EºZn2+/ Zn = – 0.76V,
EºMg2+/ Mg = – 2.38 V
Answer the following questions:
- What is the standard electrode potential?
Ans: The potential difference between a given half-cell and the standard hydrogen electrode (SHE), when all components are in their standard states (1 M concentration, 1 atm pressure, 25°C or 298 K), is called the standard electrode potential. - Construct the galvanic cell notation for the given electrodes, indicating the anode and cathode.
Ans: Anode = Mg2+/ Mg
Cathode = Zn2+/ Zn
Galvanic cell: Mg(s)/Mg2+(aq.)//Zn2+(aq.)/Zn(s) - Write the complete cell reaction for the given cell.
Ans:
At anode: Mg → Mg2+ + 2e–
At cathode: Zn2+ + 2e– → Zn
Complete reaction: Mg + Zn2+ → Mg2+ + Zn - Calculate the cell potential for the given cell at 25ºC.
Ans: Eocell = Eocathode – Eoanode = -0.76 – (- 2.38) = +1.62V
14) A chemical reaction occurs as follows:
\underset{(A)}{CH_{2}CH_{2}CH_{2}Br}+\underset{aq.}{KOH}\rightarrow B+KBra) Identify the compound ‘B’ in the above reaction.
B=\underset{Propan-1-ol}{CH_{3}CH_{2}CH_{2}OH}b) Which substitution reaction mechanism occurs in the above reaction and why?
Ans: Bimolecular nucleophilic substitution reaction (SN2) mechanism occurs because it’s a 1° alkyl halide and OH⁻ is a strong nucleophile.
c) What is the product formed when alc. KOH solution is used instead of aq. KOH in the above reaction?
CH2CH2CH2Br + alc. KOH → CH3-CH=CH2 (Propene) + KBr + H2O
d) How can you obtain nitropropane from compound ‘A’?
CH2CH2CH2Br + alc. AgNO2 → CH2CH2CH2NO2(1-Nitropropane) + alc. AgBr
15. A list of compounds is given as follows:
P-aminoazobenze, Benzenediazonium chloride, Aniline, Nitrobenzene, Phenol and Benzene
From the above list of compounds, prepare a sequence of reaction chain with suitable conditions and reagents.


‘OR’
An aliphatic compound (A) reacts with SOCI2 to give (B). The compound (B) is heated with ammonia to produce (C). The compound (C) is further heated with Br2/KOH to yield (D). The compound (D) gives (E) when treated with NaNO2 /HCI at low temperature. The compound (E) is a primary alcohol which gives positive iodoform test. Identify compounds (A) to (E) with the reactions involved.

Ans:


16. Write the chemical equations to illustrate the following reactions:
a. 2,4-DNP test
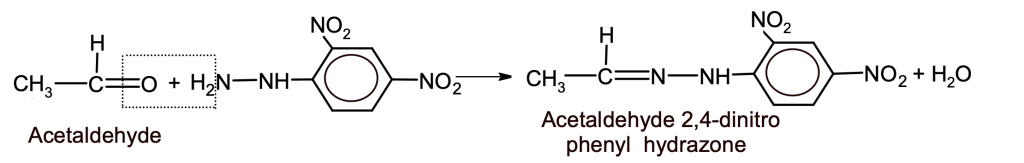
b. Reimer-Tiemann Reaction

c. Williamson’s reaction

d. Perkin Condensation reaction

e. Wurtz-Fitting reaction

17. You are given the mixture of two compounds A and B.
A = CH3CH2CH2NH2 and B = CH3CH2NHCH3
How would you separate their mixture by applying Hoffmann’s method ? Give the nitrous acid test to distinguish between compounds A and B.
Ans: Hoffman’s method of separation of primary and secondary amine:
CH3CH2CH2NH2
Propan-1-amine (Primary amine)
CH3CH2NHCH3
N-methyl etanamine (secondary amine)
1. Treatment with diethyl oxalate
a. Primary amine reacts with diethyl oxalate to form oxamide which is solid.

b. Secondary amine reacts with diethyl oxalate to form oxamic ester which is liquid.

2. Separation of oxamide and oxamic ester: Solid oxamide and liquid oxamic ester are separated by filtration.
3. Regeneration of primary and secondary amine: Oxamide and oxamic ester are separately boiled with KOH to regenerate respective amine.

Nitrous acid test to distinguish A and B:
i. A reacts with nitrous acid to form alcohol with the effervescence of nitrogen gas.

ii. B reacts with nitrous acid to form nitroso-amine, which is a yellow liquid.

18. The metal ‘M’ with electronic configuration [Ar] 3d104s2 belongs to group IIB in the periodic table and is commonly called ‘Jasta’.
a) Identify metal ‘M’ and write its main ore.
Ans: Metal (M) = Zinc (Zn)
main ore = Zinc Blende (ZnS)
b) Why is metal ‘M’ not considered a transition element?
Ans: Zinc is not considered a transition element due to the following reasons;
- Zinc forms colourless compounds, e.g., ZnSO4, ZnCl2, etc.
- Zinc does not show variable valency and oxidation state. (Only +2 oxidation state).
- Its d-electrons are not involved in metallic bonding. It possesses a weak metallic bond.
- Its d- orbital is completely filled and does not show the catalytic property.
c) Draw the vertical retort. Write its reduction reaction.

Reduction reaction: ZnO + C → Zn + CO
d) What happens when metal ‘M’ is exposed to air for a long time?
Ans: When zinc is exposed to air for a long time, basic zinc carbonate is formed.
2Zn + H2O + O2 + CO2 → ZnCO3.Zn(OH)2 (Basic Zinc Carbonate)
19. What is rusting? Describe the electrochemical theory of the rusting of iron. List any two methods for the prevention of rusting.
Ans: When iron is exposed to moist air for a long time, a reddish-brown layer of hydrated ferric oxide is formed on the surface of iron, which is called rust, and the process of forming rust is called rusting.
4Fe+ 3O_{2} + 2xH_{2}O\rightarrow \underset{\underset{(Rust)}{Hydrated\ ferric\ oxide}}{2Fe_{2}O_{3}.xH_{2}O}Electrochemical theory of rusting of iron
This is modern theory. According to this theory, when impure iron comes in contact with water containing dissolved oxygen and carbon dioxide, a galvanic cell is set up. Iron acts as the anode, and the impurities act as the cathode. Water containing dissolved oxygen and carbon dioxide acts as an electrolyte.
At the anode, the Fe atom passes into solution as a ferrous ion.
Fe → Fe++ + 2e–
At the cathode, electrons are used to form hydroxide ions.
2H2O + O2 + 4e– → 4OH–
Fe++ obtained from the anode and OH– obtained from the cathode combine to form ferrous hydroxide.
Fe++ + 2OH– → Fe(OH)2
In excess of oxygen and water, ferrous hydroxide is oxidized to ferric hydroxide.
4Fe(OH)2 + O2 + 2H2O → 4Fe(OH)3
Ferric hydroxide is unstable and loses water to give hydrated ferric oxide.
2Fe(OH)3 → Fe2O3 + 3H2O
Ferric oxide absorbs moisture from the air and gives hydrated ferric oxide, known as rust.
Fe_{2}O_{3} + xH_{2}O \rightarrow \underset{Rust}{Fe_{2}O_{3}.xH_{2}O}Prevention of rusting
1. Protective coating
- A coating of oil and grease
- A thin coating of paints and enamels
- By passing steam on red-hot iron
2. Application of corrosion inhibitor: Treating iron with certain solutions like potassium chromate, phosphoric acid, and conc. Nitric acid inhibits rusting.
3. Galvanization (cathodic protection): The process of coating zinc (electropositive metal) over the surface of iron is called galvanization.
Zinc becomes the anode, and iron becomes the cathode. The anode sacrifices itself and prevents the cathode from rusting.
4. Allowing: Iron can be prevented from rusting by allowing it to be coated with chromium, nickel, etc.


